Release date: 2014-05-14
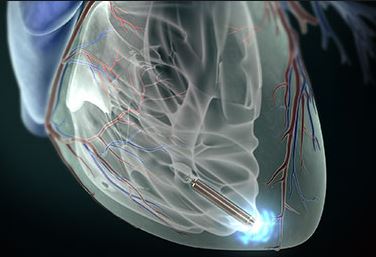
The LEADLESS study, published at the 2014 American Heart Rhythm Society Annual Meeting, showed that the 1-year follow-up of the Nanostim wireless pacemaker showed good safety and efficacy.
Dr. Vivek Y. Reddy (Mount Sinai Hospital) said that a single-chamber Nanostim wireless pacemaker placed through a catheter is designed to reduce surgical complications, which are usually caused by wires.
Three months of data (33 cases) of the LEADLESS pilot study were published in March. Reddy now reports 1 year of data. The patient was placed in the device from December 2012 to April 2013. The mean pacing threshold (0.4ms) was 0.8V when placed, 0.46V at 3 months, 0.4V at 9 months, and 0.43V at 1 year. The R wave is perceived to be 8.26 mV at the time of insertion, 10.65 mV at 3 months, 10.64 mV at 6 months, and 10.32 mV at 1 year.
90-day data showed a 97% success rate, with 94% of patients without complications. The device worked well for 90 days and 1 year without additional complications, including equipment migration or odor, ventricular arrhythmia, infection, equipment failure or early battery loss.
In February, Nanostim received the CE mark, LEADLESS II has begun to be included in the patient, and 670 patients will be included as a basis for submission to the FDA for approval. Reddy pointed out that LEADLESS placement failures include cardiac perforation, which has been reported in Europe. As with any other new technology, wireless pacemaker placement is also risky.
Source: Medical Pulse
JIANGSU CLS TECHNOLOGY CORP.LTD , https://www.js-cls.com