Release date: 2016-03-17
Medtronic recently announced that the US Food and Drug Administration (FDA) has approved Medtronic Brain Deep Electrical Stimulation (DBS) therapy for the treatment of Parkinson's disease, which has been diseased for at least 4 years and begins to develop motor complications or motor complications. patient. In 2002, the FDA first approved Medtronic brain deep electrical stimulation therapy for the treatment of advanced patients with Parkinson's disease. The therapy has been shown to have positive clinical performance in improving exercise complications, quality of life, daily activity, and reducing drug use in patients with Parkinson's disease.
Exercise complications are caused by Parkinson's disease progression and levodopa drug side effects, which have a great impact on the social and psychological aspects of patients, and their impact on daily life is usually very serious. As the quality of life of patients and caregivers deteriorates, the cost of treatment and care increases. 37% of Parkinson's patients have left their jobs prematurely, 6 months earlier. In the United States, Parkinson's disease consumes at least $14.4 billion a year, and it is expected that by 2040, there will be more than twice as many patients with Parkinson's disease in the United States.
"Strong clinical evidence suggests that Medtronic Brain Deep Electrical Stimulation (DBS) therapy improves symptoms in patients with Parkinson's disease, where early exercise fluctuations and dyskonia are not well controlled, compared with best drug therapy (BMT). The possibility is higher. Previously, the therapy was usually considered only when the symptoms of the disease had seriously affected the quality of life of the patient,†said Dr. Mahlon DeLong, a professor of neurology at Emory University School of Medicine. “This FDA decision is significant, meaning that we can consider using Medtronic deep brain stimulation before the patient's symptoms become severe. Parkinson's disease patients should be referred to experienced deep brain stimulation. A multidisciplinary center for comprehensive sensory fitness assessments, and for patients who are still socially or workable, the therapy can help improve symptoms, improve quality of life, and reduce the overall financial burden of the disease."
The FDA approval was based on data from the EARLYSTIM clinical study, which was published in the 2013 New England Journal of Medicine. The study found that patients with Parkinson's disease who used Medtronic deep brain stimulation (DBS) combined with drug therapy (BMT) improved disease-related quality of life by an average of 26% two years later; patients treated with BMT alone decreased by 1%. . In terms of long-term motor complications, the quality of life of patients receiving DBS therapy improved by 20% compared with baseline at 6 months; there was no improvement with BMT alone.
"Parkinson's disease is progressive, and the quality of life of patients will gradually deteriorate over time. The FDA's approval is important because it expands the treatment window for patients who can receive DBS therapy," Vice President of Medtronic Dr. Lothar Krinke, head of the brain regulation business of the Restorative Therapy Group, said. “DBS therapy has proven to be effective for a long time and can now be used early in the treatment cycle, providing an additional option for patients with early motor complications to maintain or restore quality of life.â€
Source: Sina
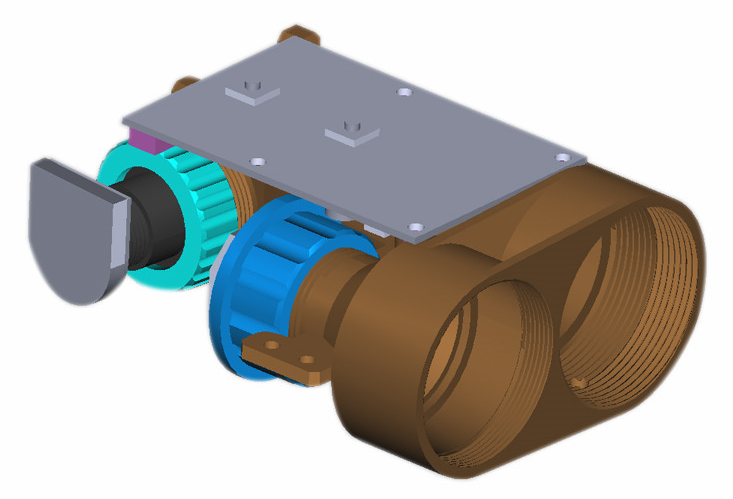
Laser rangefinder module, which has two application, one is mainly designed for long distance range finder, another is for secondary development industrial Laser Distance Sensor, which support ttl/usb/rs232/rs485 adapter output data.
With different range measuring program, JRT laser measure tools module, can satisfy customers` different requirements, 200m, 300m, 500m, 1000m, 1200m, 1500m, 3000m.
We have been in this line for 10 years, with a strong R&D ability and hard working, we are now a leading supplier of Laser Distance Meter modules in China.
Product Specifications:
|
Measuring Range (without Reflection) |
5-500m/5-1000m/5-1500m/5-3000m |
|
Accuracy |
±1 m |
|
Laser Class |
Class I |
|
Laser Type |
905nm |
|
Magnification |
6X |
|
Battery type |
CR2-3V |
|
Operating Temperature |
-10-50 ℃ (14-122 ℉ ) |
|
Size |
28*69*60mm |
|
Weight |
About 4g |
|
Operating Temperature |
-10-50 ℃ (14-122 ℉ ) |
|
Storage Temperature |
-25~60 ℃ (-13~140 ℉) |
Laser Rangefinder Sensor,Laser Range finder Sensor,Laser Rangefinder Module,Laser Range Finder Module
Chengdu JRT Meter Technology Co., Ltd , https://www.rangingsensor.com