Brooke M. Koshel and Sean M. McCarthy
Waters Corporation (Milford, MA, USA)
Key words
Method Conversion, ACQUITY Arc, ACQUITY QDa, Peptide
Application advantage
■Seamlessly convert peptide mapping from the Agilent 1100 Series HPLC system to the ACQUITY® ArcTM system without changing the method parameters.
â– Excellent intersystem reproducibility between different ACQUITY Arc systems.
â– Incorporate complementary mass spectral information into your regular workflow.
Waters Solutions
ACQUITY Arc System
2489 UV/Vis detector
ACQUITY QDa® Mass Detector
XBridge® BEH C18 column
Empower® 3 software
Introduction
The ACQUITY Arc system is capable of running HPLC and UHPLC separations on the same platform and is an LC platform that bridges the performance gap between HPLC and UPLC®. With Arc Multi-flow pathTM technology, users can easily switch between Stream 1 and Stream 2 for seamless method transitions or existing methods. Previous studies have shown that the ACQUITY Arc system can easily convert monoclonal antibodies to SEC-HPLC method 1 and CEX-HPLC method 2 . Both studies used flow path 1 to simulate HPLC separation, and the results of the two analyses showed that the relative retention time, peak area percentage, and resolution were almost the same between the systems. The purpose of this application note is to demonstrate the equivalence of peptide mapping analysis methods between different LC platforms.
Peptide mapping analysis has become a routine analysis in the biopharmaceutical industry, often using platform testing. Peptide maps can be used to characterize the amino acid sequence of a protein to identify the protein and characterize post-translational modifications. Peptide maps are often used for batch release testing or for gene stability testing after identification is completed during commercial conversion of the product. Peptide mapping is not a universal approach, and analysts must develop analytical methods for specific target proteins4. In order to obtain highly reproducible peptide maps, each analytical method must consider enzymatic and separation factors in order to accurately assess the results of identification and the key quality attributes (CQA) associated with stability, safety and efficacy.
Due to the difficulty of creating peptide maps for specific proteins, this application converted a 60-minute common platform method from the Agilent 1100 Series HPLC to the ACQUITY Arc system and evaluated the method conversion process. This method was then applied on two different ACQUITY Arc systems to evaluate the differences between the systems by comparing the results. Finally, in addition to optical inspection techniques, we used the ACQUITY QDa Mass Spectrometer to demonstrate how mass spectrometry can be applied to this type of analysis.
experiment
sample discription
90 μL of 10 mg/mL infliximab was treated with dithiothreitol for alkylation and alkylation with iodoacetamide. Add trypsin to digest the sample (the enzyme to substrate ratio is 1:20), then bath at 37 ° C for 18 hours.
Finally, pure TFA was added to inactivate trypsin. The final concentration of the sample after enzymatic hydrolysis was approximately 0.4 mg/mL, and no further dilution was required for direct injection analysis.
LC condition
LC system: ACQUITY Arc system with 2489 UV/Vis detector and QDa mass spectrometer, flow path 1
Agilent 1100 Series HPLC System with Quaternary Pump and DAD Detector
Expanded loop: 100 μL
Absorption wavelength: 214 nm
Sampling rate: 20 Hz
Column: XBridge BEH C18 130Å, 3.5 μm, 4.6 mm x 100 mm (part number 186003033)
XBridge BEH C18 XP 130Å, 2.5 μm, 4.6 mm x 100 mm (part number 186006039)
Column temperature: 40 °C
Mobile phase A: H2O with 0.1% (v/v) TFA
Mobile phase B: acetonitrile with 0.1% (v/v) TFA
Sample temperature: 4 °C
Injection volume: 75 μL
gradient:
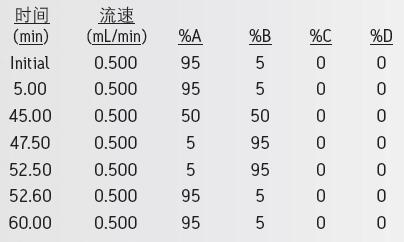
QDa settings
Sampling rate: 2 Hz
Mass range: 350 to 1250 Da
Taper voltage: 10 V
Capillary voltage: 1.5 kV
Probe temperature: 500 °C
Data management
Empower 3 CDS Software, SR2
Results and discussion
Method equivalence was achieved by HPLC peptide mapping method conversion from Agilent 1100 Series HPLC system to ACQUITY Arc system
To assess the method equivalence of the peptide mapping method from the Agilent 1100 Series HPLC system to the ACQUITY Arc system, we established a peptide map of infliximab using the method parameters described above. Based on the results of previous assessments, we are
An active preheater (CH-30A) is configured in the ACQUITY Arc system to ensure an equivalent temperature control level of both systems5. Separation was first run on an Agilent 1100 Series HPLC system to establish a baseline result (Figure 1A). Then, keep all method parameters unchanged and convert the method to the ACQUITY Arc system. The experimental results show that there is a gradient compensation volume of 185 μL between the two systems. So we applied Gradient SmartStart Technology 6 after injection to compensate for the difference in delay volume between the two systems. This function adjusts the gradient relative to the injection without changing the gradient table. The chromatogram obtained by the ACQUITY Arc system is shown in Figure 1B. The chromatograms generated by the two systems show good agreement.
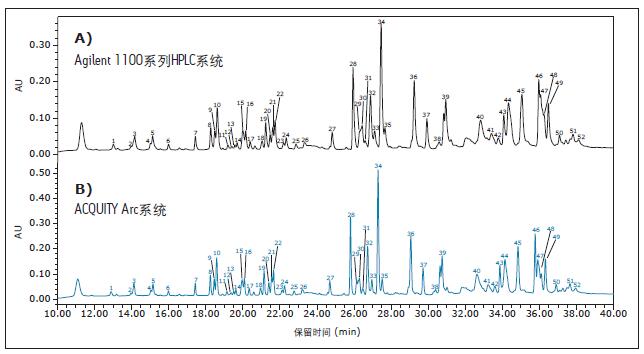
Figure 1: Figure 1. Peptide plot comparison of infliximab with data collected using A) Agilent 1100 Series HPLC system and B) gradient-compensated ACQUITY Arc system. The experiment calculated the relative retention times of all labeled peaks. The choice of peak is determined by the elution position and intensity. The relative retention time is calculated based on peak 1 .
To further assess the comparability of the results, we studied retention times. Table 1 lists the retention times of 52 peaks, two
There is a slight difference between the retention times of the corresponding chromatographic peaks obtained by the system, and the relative retention times of the corresponding chromatographic peaks
The difference is small enough to be negligible. With flow path 1 using GradientSmartStart technology, we are not changing any of the parties
The HPLC method was successfully converted to a modern LC platform on the premise of the parameters.
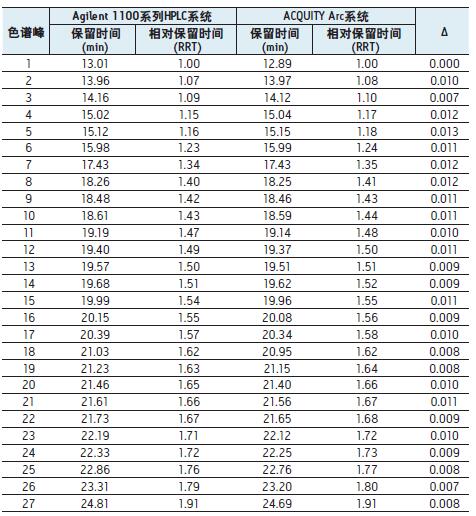
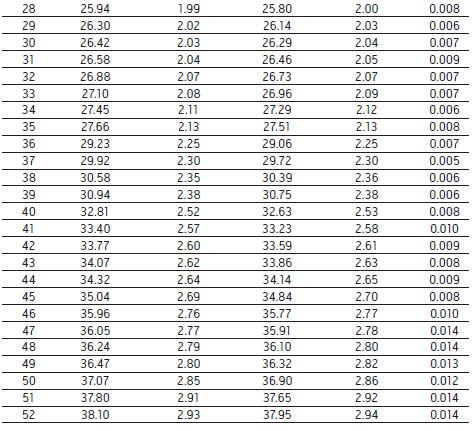
Table 1. Comparison of the retention times of the 52 chromatographic peaks identified in the Agilent 1100 Series HPLC system and the ACQUITY Arc system in Figure 1 . The Gradient SmartStart technology is used to compensate for the difference in delay volume between the two systems. Calculated
As can be seen from the _ value, the relative retention time difference caused by the compensation volume is almost negligible.
ACQUITY Arc system exhibits good intersystem repeatability when comparing two systems
When deploying new instrumentation platforms throughout the lab or other analytical sites, it is important to ensure consistent results.
To compare the analysis results of different ACQUITY Arc systems, we performed two sets of different ACQUITY Arc systems with the same core configuration, performing the analysis and collecting the data under the conditions described above. Both systems are equipped with a 30 cm CH passive preheater. The mobile phase and samples used in each system analysis were prepared separately to mimic the situation in which different analysts perform sample analysis in different laboratories in real-world industrial environments. As shown in Figure 2, the infliximab peptide maps obtained by the two systems have good agreement. After evaluating the relative retention time differences between the two systems, the relative retention time did not differ by more than 0.006 (as shown in Table 2). Almost the same results were obtained with two different instruments on the same platform, indicating the reliability of the analysis results.
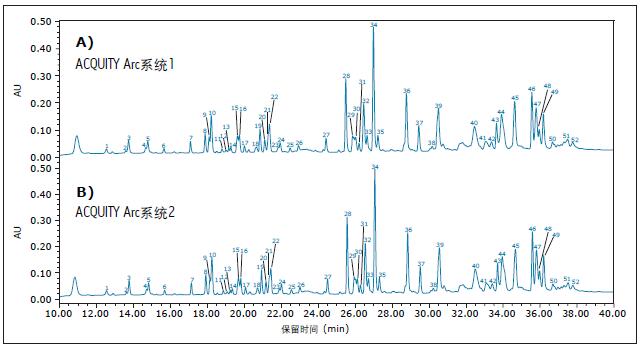
Figure 2. Comparison of infliximab peptides obtained from two different ACQUITY Arc systems: A) ACQUITY Arc System 1 and B) ACQUITY Arc System 2 . The experiment calculated the relative retention times of all labeled peaks. The choice of peak is determined by the elution position and intensity. The relative retention time is calculated based on peak 1 .
Improve the resolution of UHPLC peptide map data by mass spectrometry
In routine peptide monitoring analysis, we typically use only optical detection techniques to determine the relative retention time and peak area of ​​the sample, and then compare this data to the reference standard data to determine if the analytical results meet the fitness criteria. By introducing the ACQUITY QDa mass spectrometer as an orthogonal detection method, we can use the mass information provided to obtain more reliable product consistency analysis results. To obtain mass spectral data for the peptide mapping method described above, we connected the ACQUITY QDa mass spectrometer in series with the optical detector. This experiment used a column packed with small particle size particles to take full advantage of the ability of the ACQUITY Arc system to simultaneously run HPLC and UHPLC separations. This example replaces a 3.5 μm column with a 2.5 μm column. Instead of scaling the method for column particle size changes, we used the previous 60-minute analysis to take full advantage of the resolution and peak capacity advantages of small particle size columns.
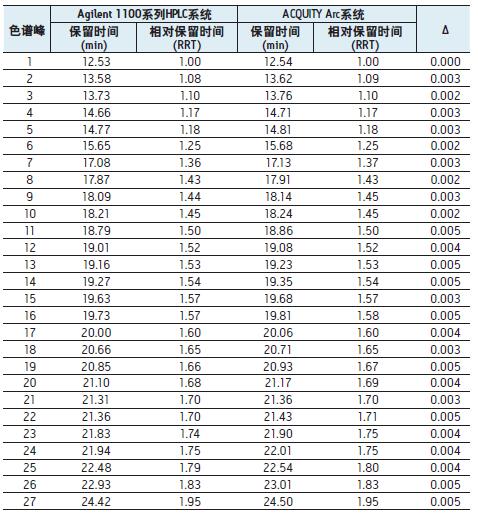
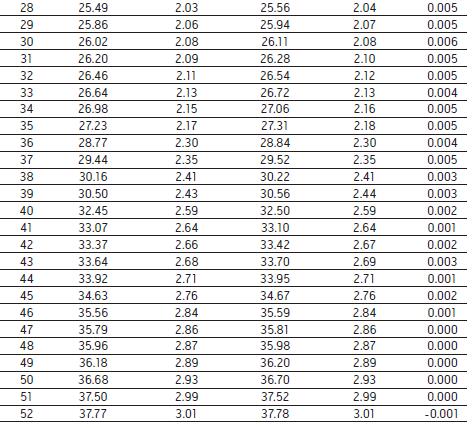
Table 2. Comparison of the retention times of the 52 peaks identified by the two different ACQUITY Arc systems in Figure 2 . The difference in retention time between systems is minimal. From the calculated _ value, the relative retention time is almost the same.
Figure 3A shows a chromatogram obtained on an Agilent 1100 Series HPLC system using a 3.5 μm column for comparison (see also: Figure 1A). Figure 3B shows the results obtained on a ACQUITY Arc system using a 2.5 μm column and Figure 3C shows the corresponding mass spectral response. By reducing the column size from 3.5 μm to 2.5 μm, small differences in resolution can be observed. The UV/Vis data is highly correlated with the corresponding mass spectral data, indicating that the ACQUITY QDa mass spectrometer can be applied to routine analysis.
The introduction of the ACQUITY QDa Mass Spectrometer provides the analyst with the mass spectral data needed to validate the identification after chromatographic peak characterization, helping them perform batch release testing and comparative analysis between batches. Taking the peptide sequence ASQFVGSSIHWYQQR as an example, the bold portion indicates the complementarity determining region (CDR) sequence. According to the average mass of the peptide 1794.0 Da, we can calculate the mass of the corresponding charge state ions [M+1H]+1, [M+2H]+2 and [M+3H]+3 are 1795.0 Da, 898.0Da and 599.0 respectively. Da. The mass spectral data of the optically integrated peak in Fig. 4A is comparable to the mass spectral data of the peak apex in Fig. 4B. We set the scan range for the ACQUITY QDa Mass Detector from 350 m/z to a maximum of 1250 m/z. From the mass spectral data of Figure 4B we can observe that different charge states can fall within this scan range. After the characterization is done in a laboratory environment, further acquisition of the mass spectrometry data can help the analyst confirm the product.
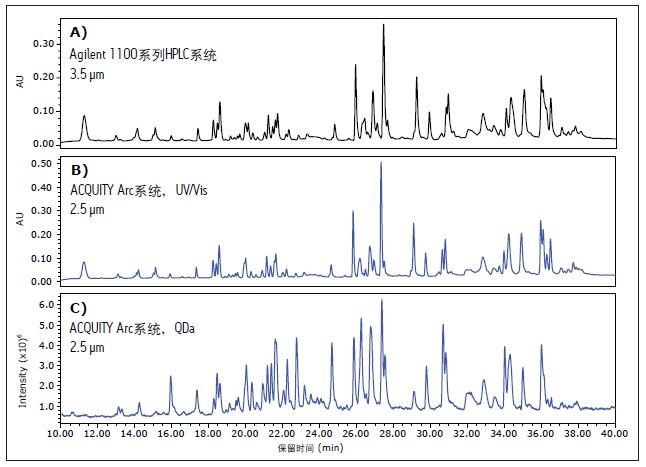
Figure 3. Peptide plots of infliximab using A) Agilent 1100 Series HPLC Systems and B) ACQUITY Arc Systems, respectively. Figure C) shows the corresponding mass spectral data obtained with the ACQUITY QDa mass spectrometer. The Agilent 1100 Series HPLC system uses data acquisition of XBridge BEH C18 130 Å, 4.6 mm x 100 mm columns packed with 3.5 μm particles , while the ACQUITY Arc system uses XBridge BEH C18 130 with 2.5 μm particles. Å, 4.6 mm x 100 mm column. The corresponding mass spectral data is highly correlated with the optical data.
![A magnified view of the infliximab peptide map from the ACQUITY Arc system showing the corresponding mass spectral data for A) optical data and B) spectrally integrated peaks, respectively. The data was collected using an XBridge BEH C18130 Å, 4.6 mm x 100 mm column packed with 2.5 μm particles. The charge states shown in Figure B) correspond to the values ​​calculated for [M+1H]+1 and [M+2H]+2 of the peptide sequence ASQFVGSSIHWYQQR (the bold portion represents the CDR sequence), respectively.](http://i.bosscdn.com/blog/20/16/77/11286193.jpg)
Figure 4. Magnification of the infliximab peptide map from the ACQUITY Arc system showing the corresponding mass spectral data for A) optical data and B) spectrally integrated peaks, respectively. The data was collected using an XBridge BEH C18130 Å, 4.6 mm x 100 mm column packed with 2.5 μm particles . The charge states shown in Figure B) correspond to the values ​​calculated for [M+1H]+1 and [M+2H]+2 of the peptide sequence ASQFVGSSIHWYQQR (the bold portion represents the CDR sequence) , respectively.
in conclusion
The biopharmaceutical industry has recognized the importance of applying new technologies and methods, and has introduced modern LC platforms such as the ACQUITY Arc system to the lab. As analytical methods become more frequently converted or converted to contract research institutions throughout the organization, we need to demonstrate that analytical methods yield consistent results on the same and different similar instrument platforms. The ACQUITY Arc system not only simulates traditional HPLC methods, but also upgrades the HPLC method to UHPLC using Arc Multi-flow path technology. By upgrading the HPLC method to the UHPLC method combined with the ACQUITY QDa mass spectrometer, we can incorporate routine mass spectrometry into the analysis to facilitate validation of the results.
references
1. Koshel, BM, McCarthy, SM Transfer of an SEC Method for Monoclonal Antibody Analysis from HPLC to UHPLC using the ACQUITY Arc System. 2015; 720005510en.
2. Koshel, BM, McCarthy, SM IEX Method Transfer: Replicating a Method for Monoclonal Antibody Analysis on an ACQUITY Arc System. 2015; 720005529en.
3. Luo, Y. et al. Handbook of Modern Pharmaceutical Analysis. In Separation Science and Technology; Ahuja, S.; Scypinski, S., Ed.; Academic Press: Burlington, MA, 2010; Vol. 10; pp 283– 359.
4. US Pharmacopeial Convention, General Chapter <1055>Biotechnology-Derived Articles- Peptide Mapping, USP38-NF33, Official from August 1, 2015.
5. Hong, P., McConville, PR Method Transfer from Agilent 1100 Series LC System to the ACQUITY UPLC H-Class System: The Effect of Temperature. 2015; 720005204en.
6. ACQUITY Arc System Brochure. Waters Brochure. 2015;720005393en.
Waters China Ltd.
Waters Technology ( Shanghai) Co., Ltd.
Beijing: 010 - 5209 3866
Shanghai: 021 - 6156 2666
Guangzhou: 020 - 2829 5999
Chengdu: 028 - 6765 3588
Hong Kong: 852 - 2964 1800
Free after-sales service hotline: 800 (400) 820 2676
Dried Broccoli,Dehydrated Broccoli,Air Dried Broccoli,Bulk Dried Broccoli,Dried Broccoli Beads
Jiangsu Tiankang Food Co., Ltd. , https://www.tiankangfood.com