For some major diseases or treatments that are helpless, such as severe liver disease, graft-versus-host disease (GvHD), renal transplant rejection, systemic lupus erythematosus, Parkinson's disease, Alzheimer's disease, etc., cell therapy has shown clear Efficacy.
In the following, Japan, South Korea will be taken as an example to introduce the development of cell therapy products in the above-mentioned regions and regulatory requirements.

1. Definition of cell therapy products and cell therapy products
Cell-based therapeutic products can be classified into adult cell and stem cell products based on cell-derived proliferative properties. Adult cell products are mostly autologous cells, stem cell products are mostly mesenchymal sources, and products are available in South Korea and Japan in Asia.
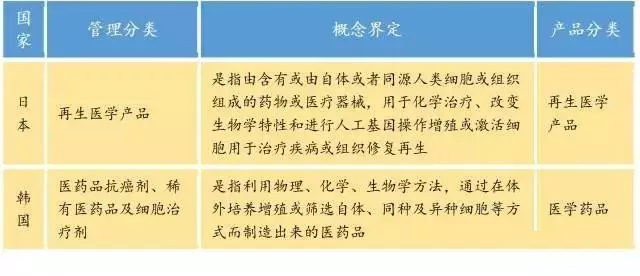
Table 1, Japan, the United States, the European Union concept definition of cell therapy products
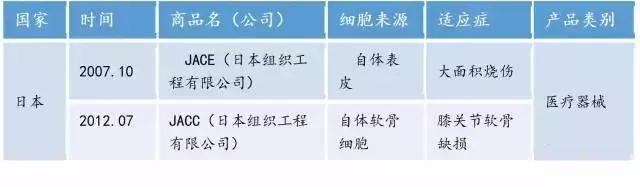
Table 2. Asian (Japan) already listed adult cell products
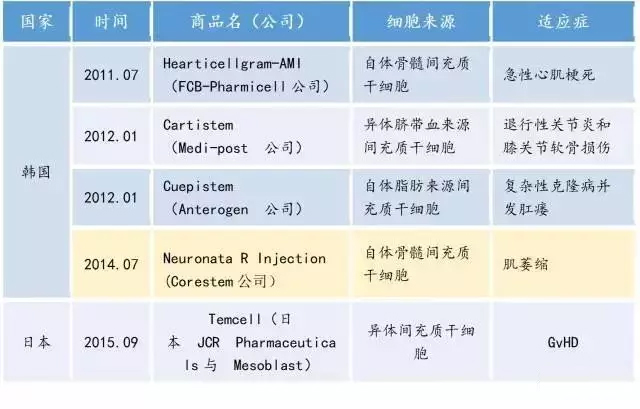
Table 3. Stem cell products already listed in Asia
Since allogeneic cells may cause rejection of the organism in clinical treatment, the risk of use is relatively high, and the therapeutic effect is highly uncertain. Most of the current clinically marketed products are autologous cell products.
Botanical Extracts (Herb extracts) refers to the ingredients extracted or processed from plant (all or a portion of the plant) by suitable solvents or methods. They can be used in the pharmaceutical, food, health, beauty and other industries.
We source and supply these ingredients of plant (botanicals) primarily to specialty food and nutritional industries. We are steadfast and committed to our sales support, research and development of any botanical we supply. We have perpetual inventory and we choose to "optimize" formulations with our clients.
Plant Extracts,Botanical Extracts,Fruit Extracts,Natural Extracts
SINOCHEM PHARMACEUTICAL CO., LTD , https://www.sinochemnutrition.com