Recently, the State Food and Drug Administration issued five announcements on medical device recalls. The announcement is as follows:
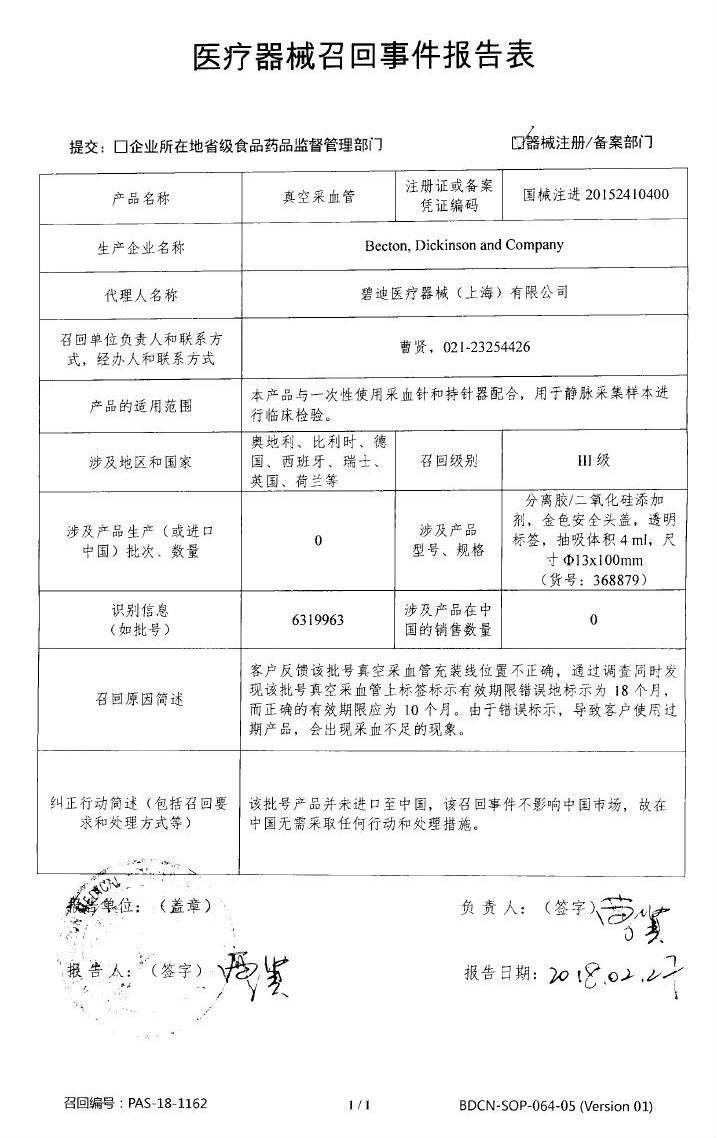
Philips Medical (Suzhou) Co., Ltd. actively recalls X-ray computed tomography equipment and X-ray computed tomography equipment
Philips Medical (Suzhou) Co., Ltd. reported that due to the possibility of free drift of the patient support device, Philips Medical (Suzhou) Co., Ltd. produced X-ray computed tomography equipment [Registration No.: National Food and Drug Administration ( The quasi-) 2013 2013 No. 3301488], X-ray computed tomography equipment [registration number: National Machinery Note 20173304046, National Food and Drug Administration (quasi) word 2014 No. 3300672] to take the initiative to recall. The recall level is three. Detailed information on the model, specifications and batches of the products can be found in the Medical Device Recall Report Form.
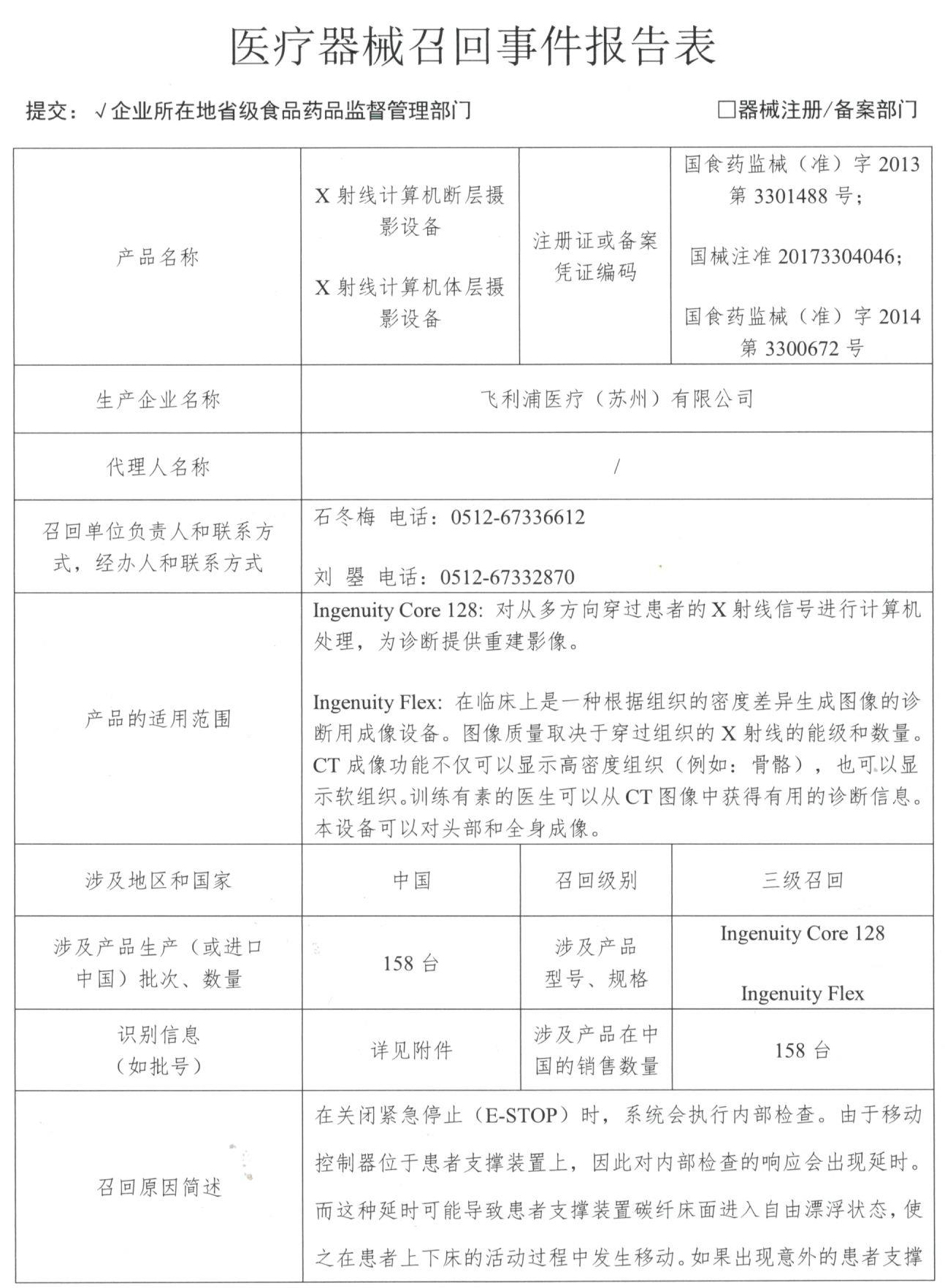
Smith & Nephew Inc takes the initiative to recall a single-use planer/grinding head/saw blade
Xerox Medical Products International Trading (Shanghai) Co., Ltd. reported that the company's single-use planer/grinding head/saw blade product uses the wrong stainless steel, and the manufacturer Smith & Nephew Inc uses it for one-time use. Planer/grinding head/saw blade (Registration No.: National Machinery Note 20153542082) Proactive recall. The recall level is three. Detailed information on the model, specifications and batches of the products can be found in the Medical Device Recall Report Form.
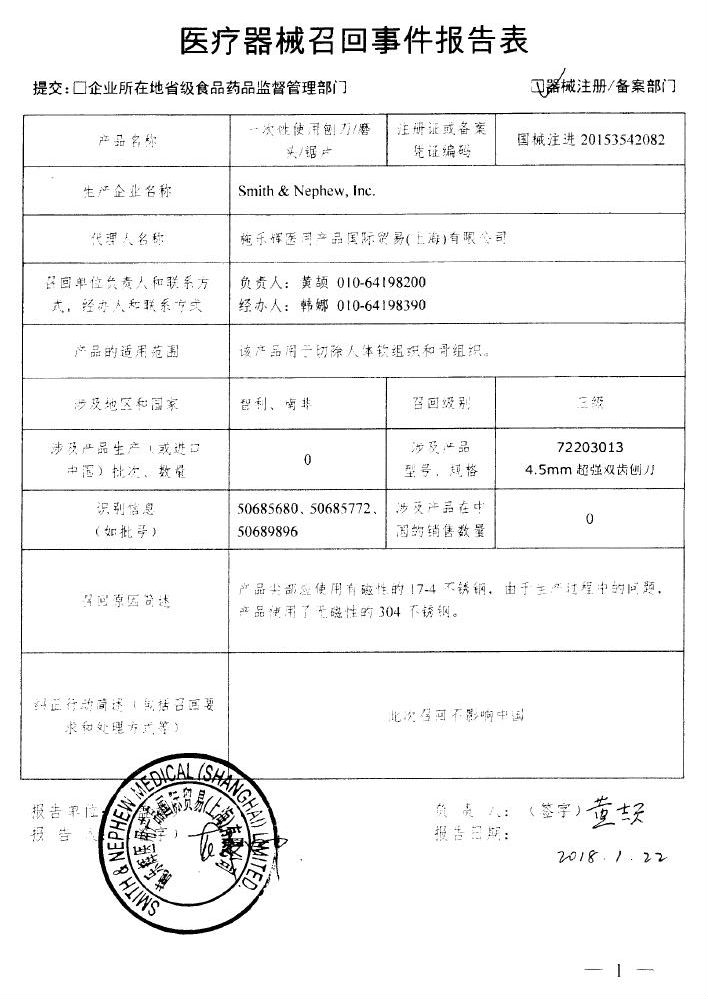
Hefei Anheng Optoelectronics Co., Ltd. actively recalls carbon dioxide laser treatment machine
Hefei Anheng Optoelectronics Co., Ltd. reported that due to the use of air-cooled lasers that did not meet the technical requirements of the products, Hefei Anheng Optoelectronics Co., Ltd. took the initiative to recall the CO2 laser treatment machine (Registration No.: National Machinery Note 20173240315). The recall level is three. Detailed information on the model, specifications and batches of the products can be found in the Medical Device Recall Report Form.
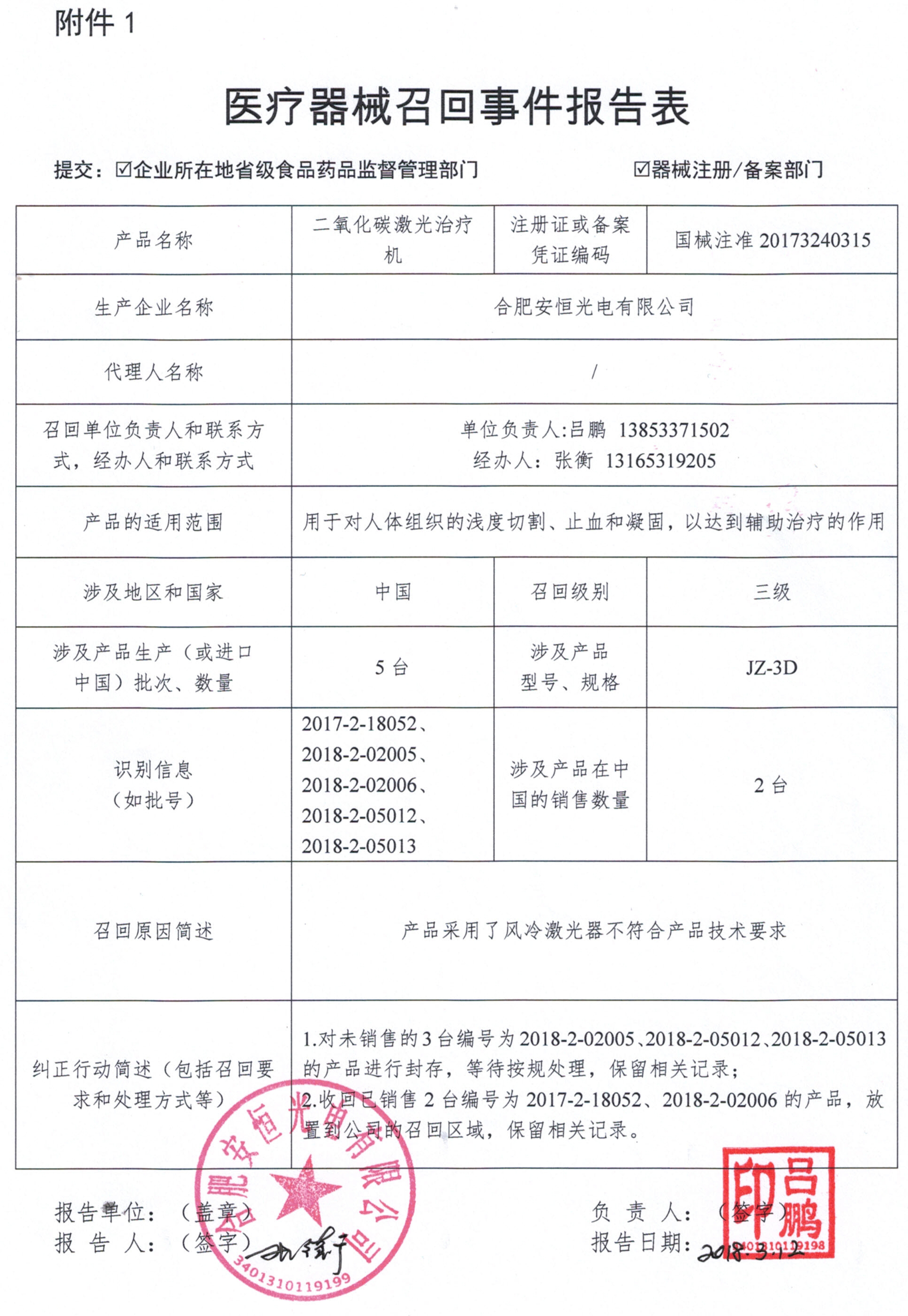
Becton Dickinson and Company voluntarily recalls vacuum blood collection tubes
BD Medical Devices (Shanghai) Co., Ltd. reported that the vacuum tube was produced by the manufacturer Becton Dickinson and Company due to the incorrect labeling period on the vacuum blood collection tube of the company's agent (Registration No.: National Machinery Note 20152410400) Proactive recall. The recall level is three. Detailed information on the model, specifications and batches of the products can be found in the Medical Device Recall Report Form.
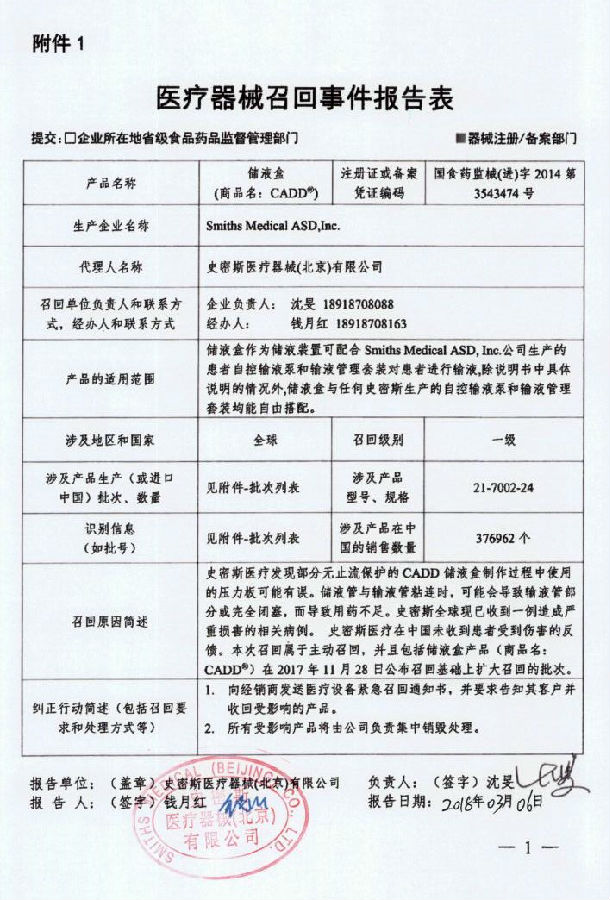
Smiths Medical ASD, Inc. Proactive recall of the liquid storage box
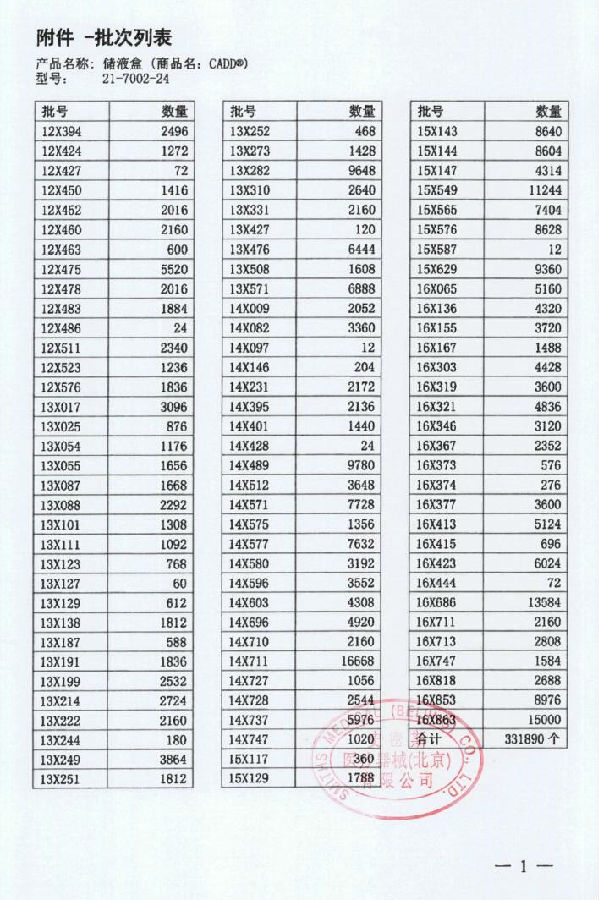
Smith Medical Devices (Beijing) Co., Ltd. reported that due to the use of the wrong pressure plate in the production process of some of the company's non-stop-protected CADD liquid storage tanks, it may cause partial or complete occlusion of the infusion tube, resulting in insufficient medication for patients. . Manufacturer Smiths Medical ASD, Inc. The liquid storage box (Registration Certificate No.: National Food and Drug Administration <Jin> 2014 No. 3543474) was actively recalled. The recall level is one level. Detailed information on the model, specifications and batches of the products can be found in the Medical Device Recall Report Form.

Lab Nitrile Gloves,Disposable Gloves,510K Disposable Gloves,Astm D6319 Examination Gloves
Puyang Linshi Medical Supplies Co., Ltd. , https://www.linshimedical.com