Release date: 2016-04-07
On March 30, 2016, US medical implant manufacturer Renovis Surgical Technologies announced that its 3D printed TeseraSC, a porous titanium cervical interbody fusion system, received FDA (510) from the US Food and Drug Administration (FDA). Market access permit. It is known that TeseraSC is a self-contained porous titanium cervical interbody fusion system with a fixed design consisting of three screws and a locking cover to prevent loose screws. The implant is used for both lordotic angles and can vary height and size depending on the situation for specific intervertebral height restorations and can be reduced by advanced instrument design.

It is understood that all Tesera implants are manufactured in 3D printing. In addition, the company currently has a patent-pending design that can produce highly porous surface structures that allow bones to grow. Deep into the implant to maximize strength, stability and stability.
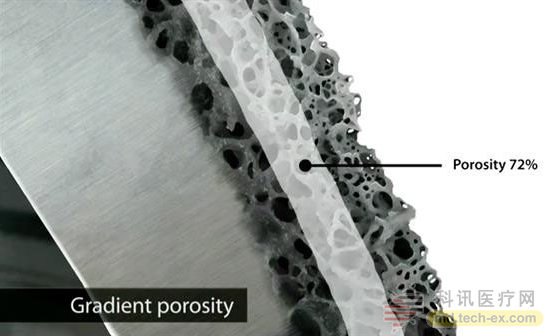
This is also the fourth FDA-approved product of Tesera's porous titanium structure from Renovis Surgical. The other three products include the independent anterior spinal fusion Tesera SA system approved in September 2013; the Tesera adult acetabular reconstruction system approved in April 2014; and the Tesera posterior lumbar intervertebral series approved in February 2015.
Source: Zhongguancun Online
We're Professional Supplier Extract Powder manufacturers and suppliers in China specialized in providing high-quality products at low price. We warmly welcome you to buy or wholesale bulk Supplier Extract Powder for sale here from our factory. For a free sample, contact us now.
Supplier Extract Powder,Supplier Extract ,Supplier Powder Manufacturer in China
Shaanxi Kang New Pharmaceutical co., Ltd. , https://www.apipepdites.com