Medical supplies are really going to drop!
Recently, the Shaanxi Provincial Health and Family Planning Commission released a message that the provinces of Hunan, Liaoning and Heilongjiang have been added to the provincial procurement alliance for high-value medical consumables. So far, the provinces that have joined the alliance have increased to 10 provinces.
This is not the only consumables procurement alliance in China. According to the statistics of Jianzhijun, the procurement alliances in the field of domestic medical consumables have: “Western Unionâ€, “Beijing-Tianjin-Hebei Procurement Allianceâ€, “Shanghai-Suzhou-Zhejiangâ€, four provinces and one city alliance and Sanming Purchasing Alliance.
In addition to the geographical relative determination of the alliance between Beijing, Tianjin and Hebei and the four provinces and cities in the east, the members of the Western Union and Sanming Purchasing Alliance are expanding and the volume of purchases is also increasing.
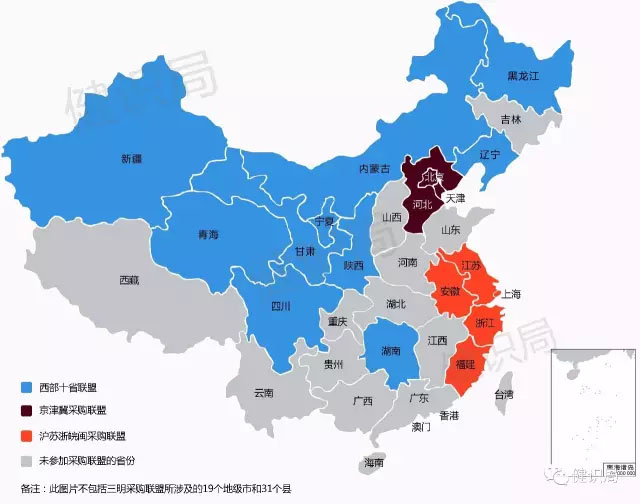
Analysts believe that the emergence of the medical consumables procurement alliance model has broken the traditional mode of medical consumables in the past, so that the purchase price of consumables is more open and transparent. After the drugs, the purchase price of medical consumables will continue to hit a new low.
High-value consumables collection documents completed before the end of October
Unlike the centralized procurement of medicines, the procurement of medical consumables is mainly based on hospitals, but the same as the implementation of provincial-level centralized procurement.
In 2013, the National Health and Family Planning Commission issued a notice on the “Regulations on the Centralized Purchasing of High-value Medical Consumables (Trial)â€, requesting the establishment of centralized procurement of high-value medical consumables on the Internet in provinces (autonomous regions and municipalities). Medical institutions and medical consumables production and operation enterprises must carry out procurement through the centralized procurement work platform established by each province, implement unified organization, unified platform and unified supervision, and mention “research and explore ways for some provinces (autonomous regions and municipalities) to jointly carry out centralized procurement†.
After the issuance of the documents, most provinces began to promote provincial-level tenders for high-value consumables. As the tenders for consumables were mostly categorized by category and batches, the provinces had different progress.
On May 5, 2017, the General Office of the State Council issued the Notice on Printing and Distributing the Key Tasks for Deepening the Medical and Health System Reform in 2017, and requested the National Health and Family Planning Commission to take the lead in formulating the guiding documents for the pilot of centralized procurement of high-value medical consumables. ", and will be completed by the end of October.
Now it seems that not only provincial-level centralized procurement, but also inter-provincial alliances have come.
Barriers to centralized procurement are being ruled out
According to industry insiders, Jianzhi Jun said that medical consumables are unique and unique. For example, the use of 7-layer masks and 23-layer masks is completely different, which is also an important reason for the difficulty in implementing unified procurement of consumables.
However, from the current point of view, this obstacle is also being ruled out. In 2016, the State Food and Drug Administration indicated that in 2016, the medical equipment classification catalogue will be re-architected, a new edition of the “Medical Devices Classification Catalogue†will be released, and a guideline for the general name of medical equipment will be organized and combined with the catalogue revision and promoted. Medical equipment coding work, tracking the progress of the US FDA, EU, IMDRF and other medical device coding (UDI) work, formulating the coding rules and coding standards, guidelines and other technical documents for the unique identification of medical equipment, for high-risk implantable medical equipment The field piloted the foundation of the unified coding work.
In April 2016, the State Food and Drug Administration also issued the “General Names for Medical Devices†to solve the problems of relatively confusing medical names, misleading identification, and exaggerated absolute terms. After the implementation of the "National Rules", the State Food and Drug Administration will organize the research on the core words and characteristics of medical device products in the "13th Five-Year Plan" period, based on the medical device product structure, technical characteristics and intended purpose, combined with the medical device classification catalogue. Words, develop guidelines for publishing naming terms, build a generic name database, and gradually promote the implementation of generic names for medical devices.
We're Professional Supplier Extract Powder manufacturers and suppliers in China specialized in providing high-quality products at low price. We warmly welcome you to buy or wholesale bulk Supplier Extract Powder for sale here from our factory. For a free sample, contact us now.
Supplier Extract Powder,Supplier Extract ,Supplier Powder Manufacturer in China
Shaanxi Kang New Pharmaceutical co., Ltd. , https://www.kangnewpharmas.com